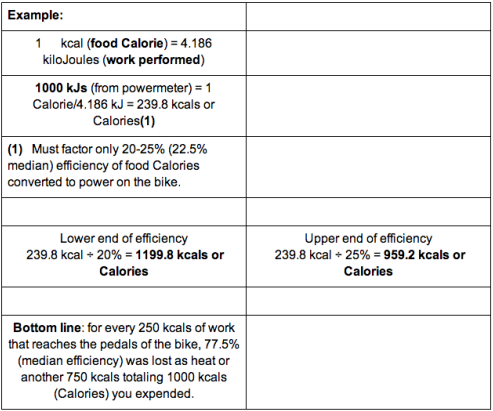
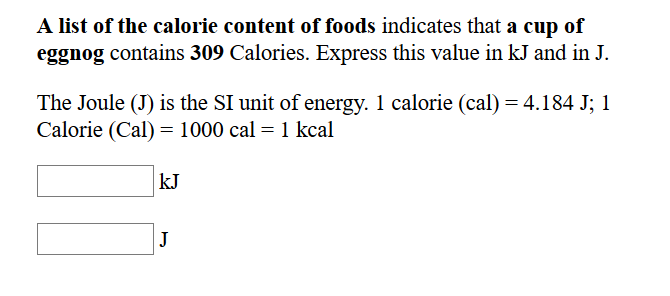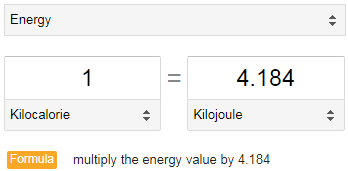Is the calorie count wrong? With the Coca-Cola Light, the calorie specification should be correct, but with the Zero variant, 250ml is specified as 2.3kJ with 1kcal. However, the conversion factor is

5.03 Conversion of kilojoules to calories | Year 11 Maths | WACE 11 Essential Mathematics - 2020 Edition | Mathspace

Chapter 6. Calorie One calorie expresses the quantity of heat necessary to raise the temperature of 1 g of water by 1° Celsius. Kilocalorie (kCal) - ppt download

Formulas to be used - CONVERSION FACTORS Calorie-Kcal 1 Nutritional Calorie, 1 Cal = 1,000 cal = 1 - Studocu
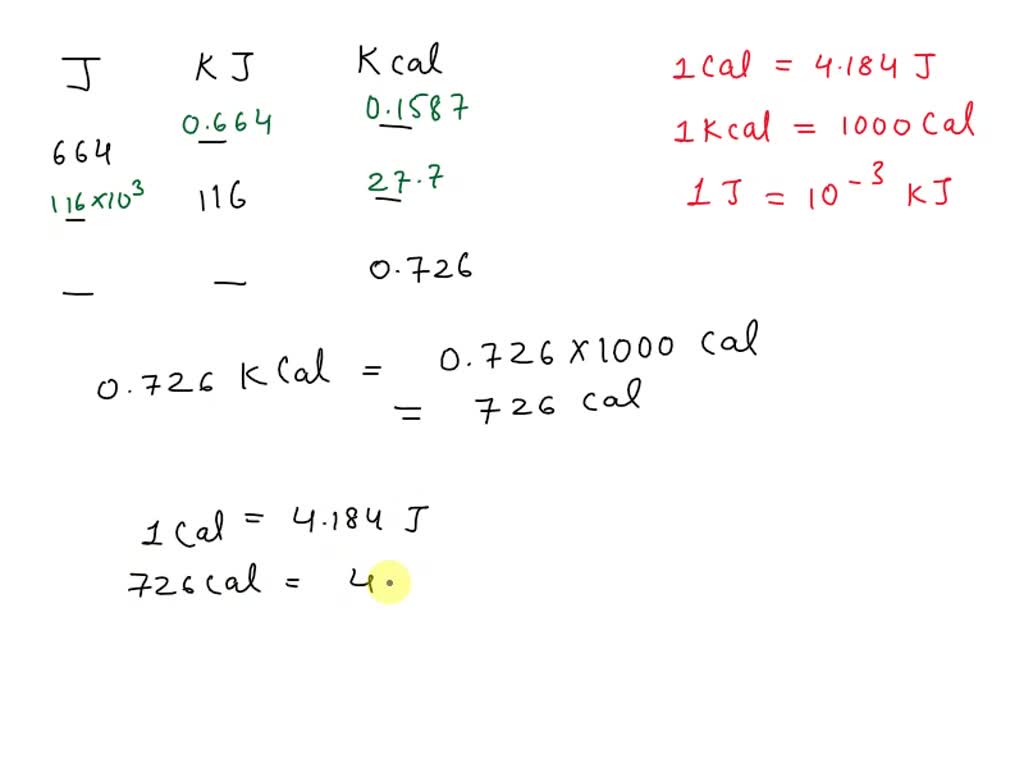
SOLVED: Expressing amounts of energy in different energy units is necessary to solve many chemistry problems. For practice, complete the following table. The Joule (J) is the SI unit of energy. 1
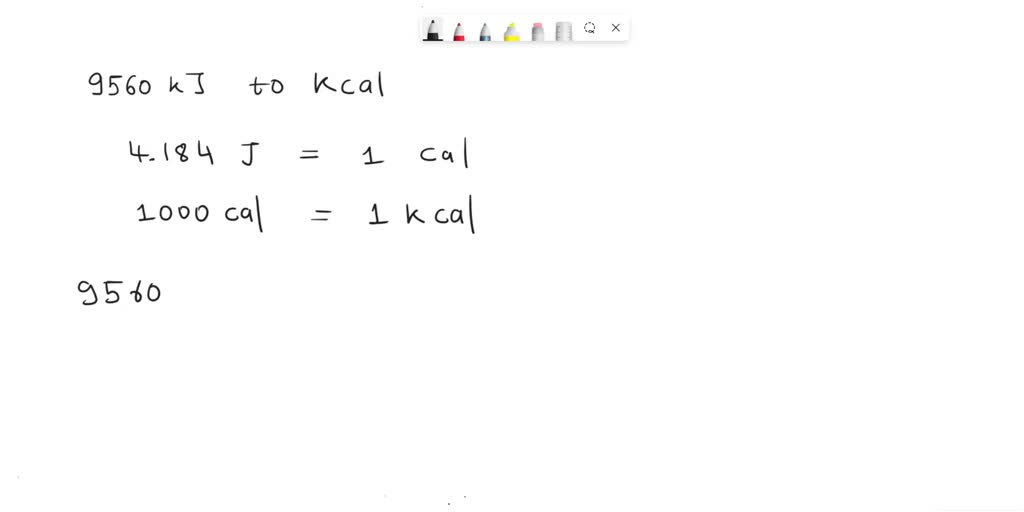
SOLVED: When 100.0 g of gasoline undergoes combustion, 9560 kJ of energy is released (Given: 4.184 kJ = 1 kcal). The heat released in kilocalories is 228 kcal.
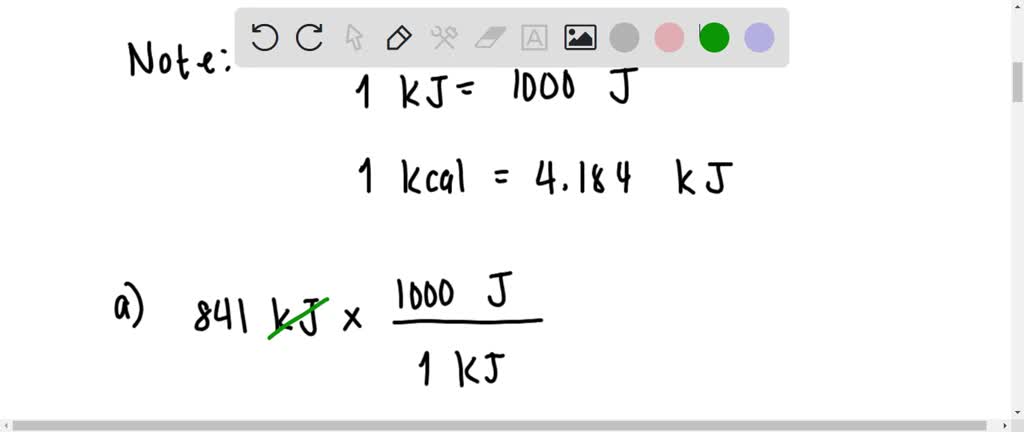
SOLVED: Perform the conversions between energy units. 841 kJ = J 2475 kcal = kJ 9.06 × 10 ^6 J = kcal